Research Collaborations
Sharon J. Nieter Burgmayer, Inorganic and Bioinorganic Chemistry
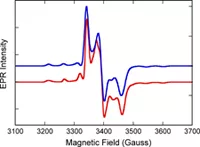
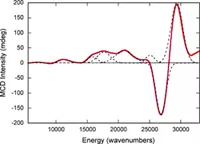
The Burgmayer labs collaborates with several labs around the country. For the project on modeling the catalytic site of the molybdenum enzymes, we have a long-standing collaboration with the specroscopy lab of Martin L Kirk at the University fo New Mexico. We provide the Kirk lab with Mo model compounds that they probe with a variety of methods: resonance Raman, magnetic circular dichroism (MCD), multi-frequency electron paramagnetic resonance (EPR). The results of these experiments combine with theoretical calculations usingi densit functional theory allows the determination of the electronic structure of the molecules. The project involving the DNA-binding ruthenium complexes has recently involved collaborator Liliya Yatsunyk at Swarthmore College who is investigating the ability of these Ru complexes to bind to quadruplex DNA. Both the Mo and the Ru projects require structure determination by x-ray crystallography which is accomplished by collaboration with Dr. Pat Carroll at U. Pennsylvania or with Dr. Glenn Yap at the U. Delaware.
Michelle M. Francl, Computational and Theoretical Physical Chemistry
I have been working in the archives at the Chemical Heritage Foundation, in downtown Philadelphia, since the Fall of 2011, in close collaboration with the staff of the Foundation. These archives include a rich trove of correspondence between scientists, particularly letters to the editor of journals and responses by the authors of the original publications to reader commentary. The goal of this research is to explore the ways in which chemists “close the loop” on publications in research journals, particularly the ways in which reader commentary in these letters to the editor affects the course of subsequent research in the field.
Jonas I. Goldsmith, Inorganic and Physical Chemistry
We collaborate with the research group of Professor Alan T. Johnson in the Department of Physics at the University of Pennsylvania. Professor Johnson’s research focuses on investigating energy and charge transport phenomena in nanoscale systems including graphene sheets and carbon nanotubes. Recently we have worked together to functionalize carbon nanotube-based transistors with large aromatic molecules, in order to examine the impact that changes in the structure of these molecules have on the transport properties of the nanoelectronic devices. Learn more about this research here.
Yan Kung, Biological Chemistry
My research into the structural characterization and manipulation of enzymes involved in the biosynthesis of compounds such as drugs and biofuels is performed in collaboration with other institutions. The bulk of the data used in my research is collected at synchrotron X-ray radiation facilities at national laboratories across the United States. In particular, X-ray data is collected as part of the Northeast Collaborative Access Team (NECAT) at the Advanced Photon Source (APS) at Argonne National Laboratory in Illinois. X-ray data collection is also carried out in collaboration with Christine M. Phillips-Piro (Franklin and Marshall College). Additional research projects on the structural investigation of biofuel-producing enzymes are performed in collaboration with the laboratory of Jay D. Keasling (University of California, Berkeley and Lawrence Berkeley National Laboratory).
William Malachowski, Organic and Medicinal Chemistry
We currently collaborate on anti-cancer research with biologists at Lankenau Institute of Medical Research (LIMR) in Wynnewood, Pa. Since 2004 we have been developing more potent enzyme inhibitors for a cancer target that was identified by our biology collaborators, George Prendergast and Alexander Muller. The target, indoleamine 2,3-dioxygenase-1 (IDO-1) and its isoform, IDO-2, are involved in the immunosuppressive activity common to all forms of cancer. Together we have developed several classes of inhibitors that are quite potent and we continue to work on new structural classes with the goal of developing a potent, selective, and bioavailable inhibitor that can be used in cancer therapy. Our work has been awarded two grants from the National Institutes of Health and has yielded several publications and patents. In fact, a biotechnology company called NewLink Genetics has licensed the technology and is currently testing it in the clinic as a new cancer therapy. More information about this project can be found here.