Susan A. White

Academic Departments
Education
Postdoctoral research, Yale University.
Ph.D., The Johns Hopkins University.
A.B., Dartmouth University.
Areas of Focus
Chemistry and biological chemistry
Biography
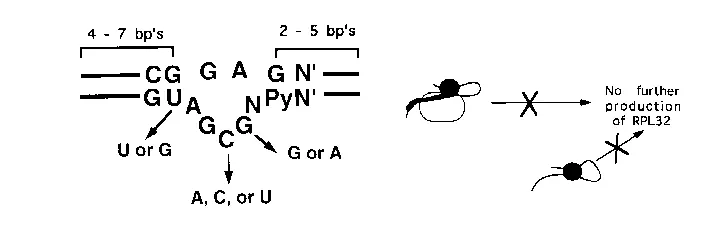
My research interests are in the area of RNA structure, stability, and protein binding. Biologically interesting RNA molecules are composed of helical regions containing standard Watson-Crick base pairs as well as internal and hairpin loops which contain unpaired nucleotides. Our goal is to understand how some of these irregular structural features contribute to the thermodynamic stability of the RNA molecule and function as sites for protein recognition. The messenger RNA for yeast ribosomal protein L30 (formerly called L32) contains an intron that must be removed before L30 can be produced. Normally, the newly produced L30 protein is immediately incorporated into new ribosomes, but when no new ribosomes are being assembled the L30 protein binds to the RNA intron ssequence. The result is that the intron is not removed from the RNA, and no additional L30 protein is produced. Several features of this feedback, or autoregulation, system are of interest. First, the RNA is composed of two rather weak helices separated by an internal loop composed of purines. Our group has verified the existence of these two helices and has shown that the internal loop is the site of protein binding (Ref 2). Furthermore, using a combinatorial approach we have shown that two GA sequences are required for protein binding (Ref 5). It is known that hydrogen bonding is possible between purines and that purines also have a strong propensity to stack on each other. Thermal denaturation experiments have shown that the purines in the internal loop are responsible for the cooperativity of the transition to the completely single-stranded state (Ref. 1). The structure of the L30 RNA:protein complex has recently been determined by NMR and supports our biochemical data (Ref 7). All but two of the internal loop nucleotides are stacked and the two remaining nucleotides jut into the protein. The RNA:protein interface is unusually intimate and we are currently making protein mutations to asses the thermodynamic contributions of the interface residues (Ref 7-9). A new crystallographic structure shows that L30 RNA forms a kink-turn structure when bound to protein (Ref. 9-10).
Publications
1. "Thermodynamic and Mutational Analysis of an RNA Purine Loop Protein Binding Site," S. A. White, H. Li, and M. A. Rauch, Journal of Biomolecular Structure and Dynamics 1995, 13, 285.
2. "Characterization of the pre-mRNA Binding Site for Yeast Ribosomal Protein L32: The Importance of a Purine-Rich Internal Loop," H. Li, S. Dalal, J. Kohler, J. Vilardell, and S. A. White, Journal of Molecular Biology 1995, 250 (4), 447.
3. "Specificity of Binding of Saccharomyces cerevisiae Ribosomal Protein L32 to Model RNAs [Symposia on RNA Biology: I. RNA-Protein Interactions, held Oct. 1995 at Research Triangle Park, NC]," S. A. White and H. Li, Nucleic Acids Symposium Series 1995, 33, 11.
4. "Yeast Ribosomal Protein L32 Recognizes an RNA G:U Juxtaposition," S. A. White and H. Li, RNA 1996, 2, 226.
5. "RNA Aptamers for Yeast Ribosomal Protein L32 have a Conserved Purine-Rich Internal Loop," H. Li and S. A. White, RNA 1997, 3, 245.
6. "Identifying critical residues at the yeast ribosomal protein L30 RNA interface" Susan A. White, Valerie Shipilov, Hongyuan Mao, and James R. Williamson, Symposia on RNA Biology III. RNA, Tool and Target held Oct. 1999 at Research Triangle Park, NC and published in Nucleic Acids Symposium Series No. 41 1999. pp. 4-7.
7. "Structure of the Yeast RPL30-autoregulatory RNA Complex Revealing a Novel Loop-loop Recognition Motif", Hongyuan Mao, Susan A. White, and James R. Williamson, Nature Structure Biology, 6, 1999, 1139-47. News and Views by A. D. Frankel "If the loop fits..." 1081-1083.
8. "A Conserved Asparagine Makes an Essential Contact to an RNA Adenosine or Cytidine",Valerie Shipilov and Susan A. White, Journal of Biomolecular Structure and Dynamics, Conversation 11 (1), 2000. 75-77.
9. "Inherent protein structural flexibility at the RNA-binding interface of L30e " Chao JA, Prasad GS, White SA, Stout CD, Williamson JR (2003). J Mol Biol. 326, 999-1004. L30e Protein Stucture
10. "Internal Loop Mutations in the Ribosomal Protein L30 Binding Site of the Yeast L30 RNA Transcript " Susan A. White, Margaret Hoeger, James J. Schweppe, Amanda Shillingford, Valerie Shipilov, and Jennifer Zarutskie, (2004) RNA, 10, 369-377. Internal Loop Mutations
11. "Compensatory Mutations in the L30e Kink-Turn RNA-Protein Complex" James J. Schweppe, Chaitanya Jain, and Susan A. White (2009), Biochimica et Biophysica Acta (BBA)- Gene Regulatory Mechanisms, 1789, 469-476.
Research Support
- NIH AREA--1993-1996
- NSF CAREER--1996-2001
- NSF POWRE--1998
- NIH AREA--2001-2004
- Mellon Foundation and AAAS/WISC sabbatical support 2002-2003
- TWAS Visiting Scientist 2004